Thank you for visiting nature.com. You are using a browser version with limited support for CSS. To obtain the best experience, we recommend you use a more up to date browser (or turn off compatibility mode in Internet Explorer). In the meantime, to ensure continued support, we are displaying the site without styles and JavaScript.
Advertisement
Nature Cell Biology (2026)
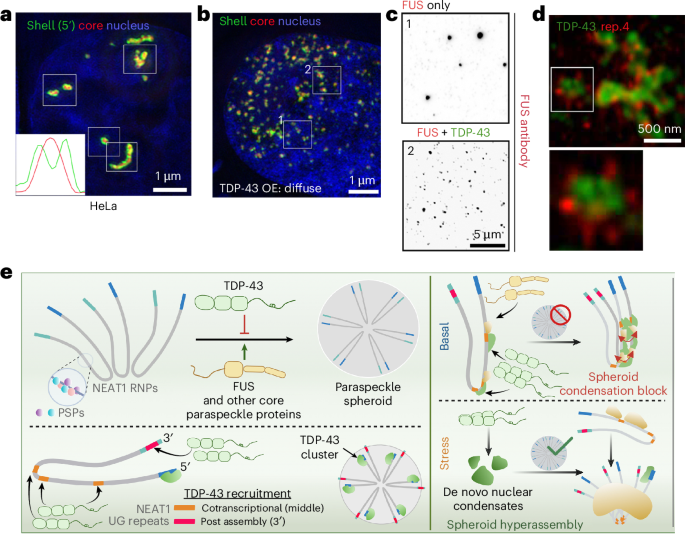
Paraspeckle condensates are membraneless nuclear organelles linked to stress responses and disease states. We demonstrate that paraspeckle assembly is tightly controlled by antagonism between TDP-43 and other paraspeckle proteins, through non-random distribution of TDP-43 binding sites on the scaffolding RNA NEAT1. Structural changes in NEAT1 or altered protein stoichiometry affect condensate formation, with implications for neurodegeneration.
This is a preview of subscription content, access via your institution
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
27,99 € / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
276,54 € per year
only 23,05 € per issue
Buy this article
39,95 €
Prices may be subject to local taxes which are calculated during checkout
Nakagawa, S., Naganuma, T., Shioi, G. & Hirose, T. Paraspeckles are subpopulation-specific nuclear bodies that are not essential in mice. J. Cell Biol. 193, 31–39 (2011). This article reported limited NEAT1_2 expression and paraspeckle assembly in most mammalian tissues.
Article CAS PubMed PubMed Central Google Scholar
West, J. A. et al. Structural, super-resolution microscopy analysis of paraspeckle nuclear body organization. J. Cell Biol. 214, 817–830 (2016). This paper describes the core-shell organization of paraspeckles.
Article CAS PubMed PubMed Central Google Scholar
Huang, W.-P. et al. Stress-induced TDP-43 nuclear condensation causes splicing loss of function and STMN2 depletion. Cell Rep. 43, 114421 (2024). This study describes TDP-43 nuclear condensation under stress and links TDP-43 oligomerization to condensate assembly.
Article CAS PubMed Google Scholar
Modic, M. et al. Cross-regulation between TDP-43 and paraspeckles promotes pluripotency-differentiation transition. Mol Cell. 74, 951–965 (2018). This study reports NEAT1 isoform regulation by TDP-43 and the role of UG repeats.
Article Google Scholar
An, H. et al. ALS-linked FUS mutations confer loss and gain of function in the nucleus by promoting excessive formation of dysfunctional paraspeckles. Acta Neuropathol. Commun. 7, 7 (2019). This paper reports defective paraspeckle assembly in an ALS subtype despite upregulation of NEAT1_2.
Article PubMed PubMed Central Google Scholar
Download references
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This is a summary of: Hodgson, R. E. et al. Paraspeckle condensation is controlled via TDP-43 polymerization and linked to neuroprotection. Nat. Cell Biol. https://doi.org/10.1038/s41556-026-01895-y (2026).
Reprints and permissions
Linking TDP-43-regulated paraspeckle condensation to neuroprotection. Nat Cell Biol (2026). https://doi.org/10.1038/s41556-026-01932-w
Download citation
Published:
Version of record:
DOI: https://doi.org/10.1038/s41556-026-01932-w
Anyone you share the following link with will be able to read this content:
Sorry, a shareable link is not currently available for this article.
Provided by the Springer Nature SharedIt content-sharing initiative
Advertisement
Nature Cell Biology (Nat Cell Biol)
ISSN 1476-4679 (online)
ISSN 1465-7392 (print)
© 2026 Springer Nature Limited
Sign up for the Nature Briefing newsletter — what matters in science, free to your inbox daily.