Thank you for visiting nature.com. You are using a browser version with limited support for CSS. To obtain the best experience, we recommend you use a more up to date browser (or turn off compatibility mode in Internet Explorer). In the meantime, to ensure continued support, we are displaying the site without styles and JavaScript.
Advertisement
npj Biological Timing and Sleep volume 3, Article number: 18 (2026)
Chronobiology has advanced scientifically since 2000. Translating this knowledge and approach to medicine can alter diagnosis, treatment, and prevention, and improve health. Adding time-of-day (or time-of-year) information is both a concrete and conceptual change to clinical practice and public health relevant to humans and other animals, with low implementation costs. Successful translation of chronobiology to medicine requires new methods, training, and organizational and regulatory action.
Basic science findings from chronobiology—the study of rhythmic processes adapting to a cyclic environment—have already had translational impacts on agriculture and are now beginning to influence medicine. Chronomedicine incorporates time-related factors into medicine, creating a multidisciplinary, interdisciplinary, and transdisciplinary field that operates at multiple levels of physiology. Chronomedicine can influence all levels of medicine, including public health and safety. It has the potential to revolutionize how we prevent disease, treat patients, and improve health. A goal over the next quarter century would therefore be to translate advances in knowledge from basic chronobiology to chronomedicine.
Some definitions are required as I proceed. “Medicine is the science and practice of caring for patients, managing the diagnosis, prognosis, prevention, treatment, palliation of their injury or disease, and promoting their health”1. Note that this definition of medicine includes (i) any animal, not just humans, (ii) all healthcare providers (e.g., physicians [MD, DO], psychologists, nurses, pharmacists, physical therapists, social workers), and (iii) health, defined as “physical and emotional well-being”2. Chronobiology studies endogenous rhythmic processes (“clocks”) that are an adaptation to environments characterized by yearly, monthly, daily, and tidal structures. Chronomedicine, therefore, would be medicine using, in its science and practice, chronobiological rhythms (i.e., yearly [e.g., seasonal affective disorder, seasonal change in pelt color or reproduction3,4], monthly/lunar, circadian, and tidal periods). Any rhythmic process could be eventually incorporated into chronomedicine provided that the rhythmic information is relevant. The period of the rhythm most studied for health-related reasons is circadian (~ 24 h)5.
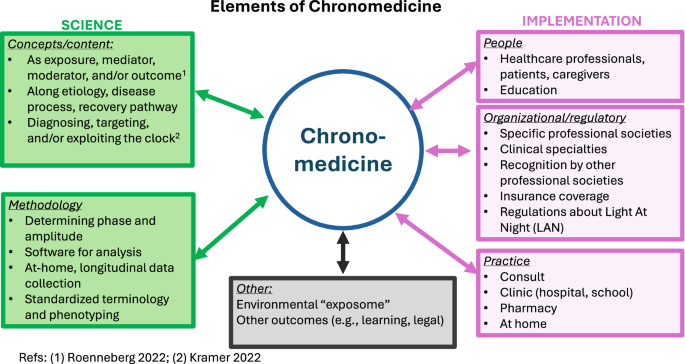
In this Perspective article, I will discuss progress over the past 25 years towards this goal, as well as current gaps in knowledge or implementation, and steps involved in achieving this goal of translating chronobiology findings to chronomedicine. Each of these is discussed within a content area: chronobiological concepts and content as applied to medicine, methodology, people, organizations/governments/regulatory, and chronomedicine practice (overview in Fig. 1). I will focus on human-based examples and on daily and yearly rhythms. These general topics also apply to veterinary medicine, to plants (e.g., agriculture6), and to other rhythmicities (e.g., lunar, tidal). Chronomedicine successes to date have primarily been within the application of chronobiologic content to medicine (e.g., timing of interventions). For successful recognition and full implementation of chronomedicine within medicine, progress in all content areas is required.
Overview of different elements within chronomedicine.
The study of circadian and other biological rhythms began with observations (e.g., plant leaf movement), added formalisms, then experimental/interventional methods, culminating in mechanistic approaches at whole animal, organ, cellular, and molecular levels. Over the past 25 years, chronomedicine followed a similar path. The recognition that circadian rhythms affected most areas of physiology has two general translational outcomes: (i) changes in drug target levels and/or metabolism across day/night meant that the same intervention given at different times would find different levels of the target7,8 (Fig. 2) and therefore have different efficacy and/or side effects, and (ii) disturbed circadian rhythms might perturb other physiology (e.g., cardiovascular, metabolism, cognitive, sleep).
Y-axis is not important. Data were collected at 6 time points (4 h apart). Lights on from ZT0 to ZT12 and lights off from ZT12 to ZT24 ( = ZT0). Open circles are Control, and Closed circles are Intervention. ** indicates significant differences between Control and Intervention at that time point. Notes: (i) Values of Control at ZT8 = Intervention at ZT0 and values of Control at ZT0 = Intervention at ZT20. (ii) Interventions at different times have different effect magnitudes, with largest at ZT4 and smallest (with no effect of the Intervention) at ZT16 and ZT20. (iii) If had only done Interventions during the dark (i.e., ZT12, ZT16, and ZT20), which is the active time for nocturnal species, the conclusion of no effect may have been made. (iv) If intervene at multiple time points with unequal number of samples/animals per time point, then the result will depend on the distribution of the timing of interventions. (v) The most efficient way to test an Intervention is to first test with a relatively small sample size at all time points, then find the time point with the largest difference between Control and Intervention, and then do the full trial at that time point.
Over the past quarter century, the conceptual understanding of circadian rhythms (as one chronobiologic periodicity) has moved from a reductionistic approach—an input (predominantly light) to the oscillator with an output—to documentation of the complexity of biological-clock function. We now know that virtually all cells, tissues, and organs have clocks that endogenously generate circadian rhythms and entrain to their environment. The suprachiasmatic nucleus (SCN) of the hypothalamus in mammals (the site of the central pacemaker) entrains to the external environment primarily via light, while the peripheral clocks primarily entrain to internal stimuli (e.g., changes in blood concentrations of substances after eating). These clocks are complex inter-related dynamic (i.e., time varying) systems with multiple feedbacks, some of which are caused by behaviors (e.g., sleeping, eating). There are also multiple time-scales involved in the circadian response: for example, prior light history over multiple days-to-months can affect circadian rhythms in birds, rodents, and humans3,9,10,11,12. This conceptual expansion increases the number and types of targets for chronomedicine intervention.
As a first step, several hypothesis related to chronomedicine needed to be formally stated13: (i) circadian rhythmicity is important for health and disease; (ii) disruption of the circadian system can cause pathologies; (iii) symptoms and signs of disease vary across 24 h; and (iv) circadian rhythms are disturbed in some diseases/disorders. These hypotheses have been tested (and continue to be tested), generating additional data as part of the scientific process of hypotheses-experiments-new hypotheses. Conceptual frameworks are required to organize data and then generate testable hypotheses14,15. Two papers proposing complementary conceptual frameworks for chronomedicine were published in 2022.
Roenneberg et al.16
Circadian rhythms or chronomedicine as exposure, mediator, moderator, and/or outcome within a system
Circadian rhythms effects as occurring during Etiology, Health/Disease Balance, and/or Recovery of a disease or disorder
Kramer et al.13
Three approaches within chronomedicine: Detecting the Clock, Targeting the Clock, and Exploiting the Clock
In the past 25 years, studies have included documentation of significance of time-of-day, weekday (vs weekend), or season in symptoms, signs, or outcomes. Notable examples of studies include:
Variations in medical outcomes by time-of-day. Increased risk of myocardial infarctions in the morning17, timing of labor and delivery18, symptoms of rheumatoid arthritis19, levels of cortisol20, and non-dipping hypertension21 were noted in the 20th century. In the past 25 years, other advances have included (i) timing of feeding/eating affecting metabolism or weight and cardiovascular outcomes22 and (ii) timing of intervention affecting efficacy and even overall survival (e.g., statins and night and some chemotherapy for cancers23).
Night or rotating shift work—with its misalignment of endogenous circadian phase and environmental time with associated behaviors—has negative effects on cardiovascular, metabolic, mood, and other functions. (e.g., ref. 24).
The timing of sleep/wake—which is heavily influenced by both endogenous circadian and external social factors—was increasingly recognized as important for health. In 2006, Roenneberg and colleagues defined Social Jet Lag (SJL)25 which occurs when sleep timing on work/school days predominantly reflects social factors while sleep timing on “free” days predominantly reflects the influences of an individual’s circadian clock. SJL has been linked to multiple physical disorders and unhealthy behaviors (e.g., smoking)16,26. Non-SJL types of variability in sleep/wake timing, which are associated with later circadian phase27, also have adverse health outcomes (reviewed in28, with more adverse outcomes continuing to be reported).
Daylight Saving Time (DST)—another example of misalignment between circadian clocks and external time—was recognized as causing public health, medical, and safety problems29.
Circadian clock genes were found outside the brain (e.g., ref. 30) and were shown to be causal or an associated finding (e.g., vascular physiology and/or blood pressure control31,32,33,34,35, metabolism/obesity36) in some pathologies.
Variations in hospital use or safety outcomes by week or season were documented37,38.
Seasonal affective disorder continued to be researched, with hypotheses and treatments related to timing of light.
Light at Night (LAN) was shown to have adverse effects on humans (although reviews39 note poor quality of many studies with many confounders) and other animal physiology—especially reproduction (e.g., ref. 40) and orientation/migration (e.g., ref. 41).
As recognition of potential time-of-day effects on physiology increased, basic chronobiology scientists began to inquire about how to translate their work to clinical outcomes; prompted by these questions from basic researchers, an article was written about steps required for medical use of basic science findings42. There was recognition that chronomedicine and sleep medicine overlap, but are distinct. Articles began to advocate for including time-of-day in clinical care (e.g., ref. 43), with specific examples of circadian medicine (e.g., ref. 44). Notable examples of intervention studies included:
Timing of eating related to weight loss efficacy45,46.
Recommendations about the timing of medications using steroids47, statins48, and anti-cancer agents49, anticoagulants50, vaccines51, and diuretic52 began, with a goal of increasing efficacy and reducing side effects.
Drugs developed and approved to treat people with Circadian Rhythms Sleep Wake Disorders (CRSWD): symptoms of shift-work disorder53, shift circadian rhythms of people with jet-lag54, and entrain the rhythms of blind people with non-24 h sleep-wake disorder55.
Light-dark cycles (compared to constant light levels) in Intensive Care Units (ICUs) were associated with significantly better outcomes: quicker weight gain and earlier discharge in Neonatal Intensive Care Units (NICU)56 and in some, but not all, studies in adult ICUs, less delirium, better sleep, and other outcomes (review in ref. 57).
Notably, not all chronobiologic-based interventions have improved outcomes. Multiple studies have not shown time-of-day effects on drugs to treat cancer, hypertension, and other diseases and disorders, or lighting in ICUs and other (e.g., elder care) facilities.
There are three classes of “failures” of translation of basic chronobiology to clinical practice. One is when there is no chronobiologic effect. A second is when the appropriate intervention at the appropriate time with an appropriate sample size and population has not yet been tested.
The third is based on the fact that not every observed daily rhythm is circadian58. For example, the concentrations of a large part of the proteome were reported to have ~24-hour rhythms in rodent studies, but once the animals were sleep deprived, most of these rhythms disappeared (e.g., ref. 59), indicating that many concentration changes were not reflecting circadian rhythms: the changes were probably due to sleep/wake or rest/activity state, which co-varies with—but is not identical to—circadian phase under normal conditions. To understand the difference, one needs to understand the chronobiological concept called masking (i.e., evoked changes)60. Many observed daily rhythms represent the non-additive combination of endogenous circadian rhythms in physiology and evoked physiological changes (usually sleep-wake associated behaviors, including sleep/wake state, rest/activity, eating/fasting, posture, light exposure, exercise levels, social interactions)61. Since most humans have a daily rhythm that includes sleeping at night and being awake during the day, many other behaviors and aspects of physiology, such as heart rate, general activity, and eating, demonstrate such combined daily rhythms. External factors may also affect these observed rhythmic outputs: for example, the non-uniform distribution of hospital orders across the 24 h would affect observed daily rhythms in any physiologic parameter affected by drug administration62, and specific events (e.g., holidays) may mask any endogenous seasonality37.
This misattribution of a biological signal as circadian and/or daily (evoked) rhythms results in two types of problems. One is related to identifying the appropriate underlying cause or physiology. Both circadian and evoked rhythms are highly relevant for chronomedicine, and knowing the origin of their rhythmicity (endogenous or evoked) will be important for diagnosis and therapy. Intensive inpatient study protocols are currently required to separate endogenous and evoked influences on physiology61. “Circadian rhythm” studies should only report on the effects of endogenous circadian rhythm(s). This circadian vs daily distinction is important for intervention target: circadian interventions currently include light and a few drugs (with hopefully more on the way) while interventions to change daily rhythms require behavioral change (e.g., of timing of sleep/wake, eating/fasting, exercise, or intervention) and/or some drugs. Note that since central and peripheral circadian clocks interact, manipulation of any behavior (e.g., the timing of eating, exercise, sleep) may affect peripheral circadian clocks and/or their relationship to the central circadian clock. It is possible, therefore, that some effects currently considered as masking/evoked represent peripheral oscillator outputs.
The second misattribution problem (of circadian vs daily/evoked factors) is that the timing relationship between circadian rhythms and sleep-wake cycles differs in nocturnal rodents and diurnal humans: the timing of circadian rhythms in the SCN relative to the light:dark cycle is the same in diurnal and nocturnal animals, despite their sleep-wake cycle being opposite63. Since many basic chronobiologic studies are done in nocturnal rodents, this different relationship may impair the translation of basic science studies (primarily done in rodents) to human clinical outcomes64 in any physiology—not just circadian physiology.
Several major gaps in knowledge exist:
Whether there are disorders or diseases—either currently known or not yet described—that have a chronobiology etiology/cause, but we do not yet have the tools/methods to detect them. This gap can be approached in two conceptual ways: (i) based on current knowledge of physiology, what pathophysiology would be expected if there were a mutation or abnormality? and (ii) what diseases/syndromes should be investigated to determine if there is no functional circadian (or other periodic) output and/or misalignment of the peripheral oscillators or other abnormality involved? As noted above, some “clock” genes influence non-circadian physiology, and their mutations are associated with some pathologies. For example, polymorphisms in CRY2, MTNR1b, CLOCK, and PER3 are associated with different clinical metrics associated with Type 2 diabetes (reviewed in ref. 65). Individuals born with mutations of “clock” genes would provide much useful information; the inability to “find” such individuals may be either that that mutation is fatal and/or it has limited consequences. The chronomedicine field should discuss whether these pathologies should be included in its science and practice.
Is a strong (i.e., large amplitude) rhythm always beneficial? This was recently discussed in the context of recovery from illness in an ICU66. Unfortunately, endogenous circadian amplitude has not been recorded in most, if not all, clinical measures; what has been measured are daily rhythms that reflect both endogenous circadian and other factors. More data on circadian amplitude are required to address this.
Since 2000, there have been major advances in the methodology for documenting circadian rhythms or inputs that may affect circadian and seasonal rhythms. Notable examples included:
Continued improvement in wearables that enable non-invasive longitudinal assessments of multiple variables (e.g., activity, light exposure, heart rate, skin conductance) as well as ecological momentary assessment (EMA) of subjective states (e.g., pain) and marking time of events67,68.
Major advances in deriving circadian phase in an individual’s blood or other biospecimen samples69,70,71,72.
Multiple attempts to derive circadian phase from actigraphy (i.e., light and activity data) or other ambulatory data73. Unfortunately, no methods have been shown to accurately predict circadian phase under conditions when a person is not sleeping/waking at “normal” circadian phase74. The relationship between circadian phase and sleep/wake timing depends on many variables, including sex75, age76, chronotype (e.g., “morning/lark” vs “evening/ owl)77,78, prior light history79,80,81, some disorders/diseases including total ocular blindness82,83, and self-selected behaviors (e.g., due to work schedule or personal responsibilities). Sleep/wake timing, therefore, should not be considered an accurate proxy for circadian phase13. The accuracy required may depend on reason for determining circadian phase. If clinic visits can only be scheduled morning vs. afternoon (and not within a smaller time window), then less accurate methods may be sufficient. Use of the information to shift circadian phase to a desired new phase, however, may require higher accuracy, especially near the times of transitions between phase delays to phase advances (e.g., late afternoon flights from the East Coast of the North America to Europe result in light exposure that may cause phase delays rather than advances).
Mathematical methods for calculating optimal timing over single or multiple days of the administration of drugs to achieve desired phase shifts84,85. These methods will rely on accurate close-to-real time assessment of circadian phase and possibly amplitude.
Use of pupillometry to define non-visual pupillary responses to light, with possible implications for sleep, circadian and seasonal disorders, and changes with aging86.
Methods are needed for documenting key parameters of circadian or other rhythms in patients and research participants in close-to-real time. The most commonly used parameters of rhythmic phenomena are (i) phase (i.e., timing, usually relative to a local clock time or another biological process [e.g., timing of melatonin rise relative to sleep onset]), (ii) amplitude, and (iii) period of central and peripheral oscillators. For most creatures living on a 24 h day, the period of a cycle does not need to be assessed, since their endogenous circadian cycles will be entrained to local 24.0 h cycles, and yearly/monthly/tidal periods also will not change. Therefore, I focus on methods for phase and amplitude. As noted above, multiple labs have developed biochemical and statistical methods using one or two blood samples in healthy humans to calculate circadian phase. These methods need to be validated on patient populations, in whom the relative amounts of the different components used to calculate phase may differ. For eventual increased clinical use, all methods should have results in close-to-real time: current methods using assays of biosamples (e.g., saliva, blood) must await the assay (which may be done in batches, further delaying results) to be done.
Continued development of new biological data collection methods, including “wearables” (e.g., on finger, wrist, scalp), elsewhere on the body (e.g., similar to continuous glucose monitor on the arm), or ingestible (e.g., for temperature, gut biome sampling) is needed. Monitors could include alerts for medication timing, safety, or other reasons. New diagnostic tests may require a new drug for stimulus or suppression (as in glucose tolerance testing for gestational diabetes or the dexamethasone suppression test for cortisol) of circadian rhythms. Combining data from multiple data sources and/or longitudinally may be required for accurate determination of chronomedicine metrics. Advances in computing using AI/Machine Language Learning (MLL) tools are expected to be involved in this.
Needed are data collection methods and protocols that people can do at home (e.g., ref. 87) and over weeks, months, and even years. This will enable a more diverse and probably more representative group of participants in research and increased access to diagnosis and care for patients. It will also enable a better understanding of intra-individual and inter-individual variability and their causes.
Another challenge to chronomedicine is that most studies do not include testing at all circadian phases, especially in relationship to sleep/wake timing. If the response depends on circadian phase, and if not all circadian phases are studied, then: (i) an effect may be missed and (ii) the overall effect may depend on the number of data points at each circadian phase (Fig. 2). Any unequal distribution of timing of interventions within the study protocol, therefore62, may affect outcomes—including a false negative effect. Appropriately designed experiments need to be conducted to overcome this defect in protocol design. Similar work may need to be done for yearly and monthly rhythms.
Timing of interventions to affect the circadian clock requires knowledge of each individual’s circadian phase and drug pharmacokinetics. Mathematical methods based on control theory will be helpful (e.g., refs. 84,85). Control theory uses mathematics to manage dynamic (i.e., time varying) systems to achieve desired outputs (e.g., specific phase shift) while minimizing errors and instability; it is different than predictive modeling. For example, many experiments have tested the ability of an intervention at a specific time to achieve a phase shift; some models predict that result, while control theory allows calculation of the timing and magnitude of an intervention (or several interventions) to achieve the desired phase shift. Also needed for planning any timing of intervention is a discussion about how accurate determination of phase is required, given constraints in implementation, as discussed above.
A major need is consistent definitions/terminology/nosology, and standardized phenotyping. These are required for comparisons across studies (including of different populations), meta-analyses, more efficient use of research resources, and for eventual clinical translation. The NIH PHenX toolkit (https://www.phenxtoolkit.org/) is an example of a public repository for consistent definitions/terminology that chronomedicine can use once it has decided on them, and metrics to use for phenotyping. The field does not even have consensus on its name: “circadian medicine” is used more often, but, as noted above, “chronomedicine” would include non-circadian periods, with circadian medicine as a subfield.
Databases relevant to chronomedicine (e.g., UK Biobank) should include questions(s) (e.g., about sleep;/wake timing, chronotype, recent night or shift work), samples, and biomarkers related to circadian rhythms.
Continued development of and education about appropriate statistics for chronobiology research and for applications (e.g., descriptive vs prescriptive statistics) will be required. For example, some circadian or seasonal data are longitudinal (i.e., multiple data points from an individual) and all rhythmic data require consideration of cyclical analyses (i.e., the difference between 2300 and 0100 may be 2 h, not 21). The data are frequently (pun intended) not Gaussian (“normal”) in distribution; thresholds and non-linear effects should be considered; only rarely should sinusoids be used (and after testing for appropriateness)61,88,89. Appropriate handling of missing data also requires care61.
Finally, translation of chronobiology results to chronomedicine will benefit from open access to data and validated analytic and statistical programs, such as are found on sleepdata.org, maintained by the USA NIH-supported National Sleep Research Resource.
The chronobiology field currently includes individuals with research and clinical backgrounds with genetic, molecular, biochemical, whole organism, and other approaches. Many prominent people in the field trained in other areas and then applied their knowledge to chronobiology or how chronobiology affects their primary interest.
The practice of chronomedicine will depend on healthcare professionals, patients and People with Lived Experiences (PWLE), caregivers, and advocacy groups, and their joint efforts. All groups should contribute to the development of materials and approaches used for education and training, both for themselves and for other groups. This education and training would include general interdisciplinary materials and then more group-specific materials and skills. For healthcare professionals (e.g., physicians, clinical psychologists, nurses, social workers, public health professionals, and pharmacists), basic knowledge of circadian and other physiology (and sleep) should be taught in their professional school and then clinical training, including clinical fellowships in chronomedicine (e.g., similar to cardiology or Behavioral Sleep Medicine).
Master’s level programs aimed at people with diverse backgrounds should also be established. For example, the University of Lisbon (Portugal) has a Master’s program in Sleep and Circadian Medicine.
Healthcare professionals, patients and PWLE, and advocacy groups should participate in education and outreach for the general public. Materials in different formats for this work are required. Support and recognition are required for professional and family/non-professional caregivers who must work around-the-clock to care for individuals.
Multiple organizational advances related to translating chronobiology research have already occurred in the past 25 years. Notable examples include:
The International Agency for Research on Cancer (IARC) within the World Health Organization (WHO) declared shiftwork as Group 2 A carcinogen, meaning shiftwork is ”probably carcinogenic to humans” in 200790.
Circadian rhythm sleep-wake disorders were defined (i.e., International Classification of Sleep Disorders (ICSD)), including those internal (e.g., non-24) and external (e.g., from shift-work and jet lag), first in 1990 and with definition updates in 2005 and 201491. Within sleep disorders, especially insomnias, abnormalities of the relationship between circadian rhythms and sleep timing may be present83.
Dr. Zee established the first USA Circadian Medicine clinic (“Center for Circadian and Sleep Medicine”) at Northwestern University in 2014 to translate efforts of the research-based Center for Sleep & Circadian Biology (founded 1995 and renamed in 2000).
In 2018, Dr Roenneberg organized a workshop “The Role of Circadian Biology in Preventing and Treating Pathology”. In his presentation at this workshop, Dr. Czeisler suggested the idea of a Circadian Medicine field, similar to, but distinct from, Sleep Medicine.
The International Association of Circadian Health Clinics (IACHC) was formed in the 2020s. Their vision statement is “to improve health through the development, assessment, and implementation of evidence-based, personalized care for patients with any condition or disease of the circadian system or associated with disruption of the circadian system”.
In 2024, the Society for Research on Biological Rhythms (SRBR) hosted a Circadian Medicine Continuing Medical Education Course as a supplement to their bi-annual meeting.
Organizational efforts, governmental, and other regulatory changes will be key for advancing chronomedicine. Chronomedicine will need to be recognized as a sub-specialty within each professional realm. If the model of Sleep Medicine becoming recognized as an MD specialty is followed, the steps include: establishing payment methods (e.g., insurance billing codes), creating a fellowship (with both basic science and clinical topics), writing a textbook (with both basic science and clinical topics), developing an exam, and then seeking certification from the appropriate organization. All these steps may need to be modified for the regulatory environment of each country for each clinical type (e.g., physician, psychologist, nurse, social work, pharmacist, public health).
Eventually, separate national/international annual meetings on chronomedicine may occur. In the beginning, there may be separate sessions within more basic science meetings (e.g., SRBR, European Biological Rhythms Society, Australasian Chronobiology Society, Latin American Chronobiology Group) and within other clinical specialty meetings (e.g., American Heart Association (AHA), American Public Health Association, National Association of School Nurses). Another step would be the formation of clinically-focused societies and journals, since basic science-targeted societies and journals are already established. Industry support is needed. Non-profit foundations (for public outreach, research, or junior faculty support, and other programs) should also be established.
More associations need to state that circadian rhythms are important, similar to AHA adding Sleep Health to their Essential 892 list. As more organizations make this step, the importance of the field will be better known, and there will be increased pressure for clinicians to include chronomedicine in their practice, with associated changes in insurance coverage.
Healthcare and other administrators will need to be convinced of the need to staff clinics, laboratories, and other units so that patients can be seen and treated and studies can be done outside of “normal” working hours.
As noted above, insurance coverage for methods for diagnosing (e.g., actigraphy, Dim Light Melatonin Onset) and treating chronomedicine disorders is required. This will require additional data about the cost-effectiveness of the diagnostic tool, consultant time, and/or intervention.
Interactions with industry for drugs, devices, and software and imaging tools, as well as for supporting clinical trials will be key.
Regulations about the environment, including decreasing LAN and increasing indoor light levels (and/or specific wavelengths of light) indoors during the day—including in hospitals/long-term care homes/school, and from space (e.g., satellites), on space vehicles93 or other planets—should be discussed. As noted above, LAN has effects on seasonal rhythms that affect survival, including reproduction and camouflage in non-human species.
For this human-centric author, chronomedicine is the most important translational outcome of chronobiology research. As noted above, some of this work has begun.
For physicians who focus more on abnormal physiology, the practice of chronomedicine may include both consultations (e.g., actigraphy use and interpretation, blood/saliva tests of circadian “time”, appropriate timing of an intervention) and clinic visits (e.g., CRSWDs). Treatment of CRSWDs should probably be moved to the chronomedicine field from sleep medicine since sleep is an outcome variable for those disorders, rather than the underlying cause (both endogenous [e.g., non-24 h] and exogenous [e.g., shiftwork] causes). Two different models of medical specialties that chronomedicine could follow are: (i) Interpretation of tests or other data collection that are used by other clinicians: for example, radiologists interpret MRI or CT results and communicate those results to other clinicians who then use this information for diagnosis and/or treatment. (ii) Daily practice of clinical care is informed by the additional knowledge and skills of the specialty (e.g., genetics or gerontology). Training programs may begin within Sleep Medicine because of content overlap (i.e., sleep) and to take advantage of administrative structure.
For psychologists, nurses, and social work professionals, who focus more on personal and situational factors influencing behavior and health, their chronomedicine treatment practice may include education and training in techniques about modifiable factors (including behavioral interventions) to the patient and advocating for the patient regarding situational factors.
For pharmacists, the practice may include recommendations about timing of administration of medications to improve efficacy and/or reduce side effects for patients, and education of physicians about this topic.
For public health workers, the practice may include education and public health measures (e.g., lighting environments, DST) as well as ways to increase access to care at night, especially for people with psychiatric and addictive (e.g., substance use, gambling) or violence problems94. This work is essential if we want to move from a reactionary (disease focused) society to one that invests in promoting health.
All types of practitioners should seek opportunities to consult for governments, companies, foundations, and unions or other worker’s organizations about working conditions (e.g., lighting, shift schedules) that will improve worker health and performance and reduce errors and accidents.
The details of the practice of chronomedicine will partially depend on the payment system of the locale. For the USA, government and private insurance plans may vary in their coverage. As noted above, additional data are required to convince insurance companies and/or the government to pay for testing, professional time, and interventions.
For the practice of chronomedicine, one step would be documenting a patient’s “biological time” (which may not be the same as local clock time) and/or chronotype in their medical records so that (i) interventions can be appropriately timed to increase efficacy and/or reduce side effects and/or (ii) treat patients whose biological time is desynchronized from the local clock time (e.g., jet lag) or who have problems when their multiple internal biological clock times are not synchronized. Practice changes could include adding a metric of a patient’s biological time and/or chronotype to vital signs, considering patient biological time and/or chronotype along with age or sex as a potential covariate/confounder, including patient biological time and/or chronotype when interpreting symptoms/signs or planning interventions or counseling patients, and/or recommendations about lighting inside and outside during the day and during the night. The timing of all events (e.g., sample collection time, meal time) should also be recorded61.
Exogenous factors and behaviors will also need to be documented as the practice of chronomedicine evolves. For example, many aspects of urban life (e.g., lighting conditions, sleep/wake timing, exercise, medications [prescription, non-prescription, over-the counter, recreational, supplements]) affect yearly, monthly (in some women), and daily physiology and therefore fall under the umbrella of chronomedicine. Therefore, practice approaches may incorporate consideration of both endogenous and exogenous factors, even though some may be non-modifiable.
More clinical and implementation studies need to be conducted to document the efficacy and cost-effectiveness of chronomedicine practice for the patient, their caregivers, insurers, and healthcare systems. Quality of life measures should also be incorporated into these studies.
Finally, there are multiple related topics that may benefit from advances in chronomedicine or the concept that time-of-year/month/day influences outcomes. Two examples are:
The effects of the environment (“exposome”, including biological and psychosocial factors)95. For example, people with lower-income/disadvantaged backgrounds are more likely to have early work and shift work schedules.
Effects in education/learning, legal96 and other outcomes.
In the past 25 years, advances in chronobiology enabled the goal of translation of their results to chronomedicine13,16,43 based on efforts of multiple people from many disciplines and approaches who created a strong framework of ideas, methods, and organizational structures. A final goal is a move towards Chrono-Health in which the individual’s overall health—not just absence of disease—is important. Continued chronobiology basic science results and development of methods, people, and organizational structures are needed to advance chronomedicine into all aspects of medical and health care and public policy for humans and other animals. Chronomedicine could advance the field of medicine as a whole: using time (of year/season, month, day) as an additional factor is a relatively low cost way of improving current treatment approaches without developing and testing new drugs or devices, and offering novel paths for intervention, including those at levels (e.g., hospital organization) that are not typically leveraged to improve health and well-being. This article has been written from the perspective of an individual with medical training. I have attempted to include other perspectives (e.g., nursing); others with more expertise in different approaches should also contribute to the discussion about the future of chronomedicine.
N/A. This manuscript does not report data generation or analysis.
Medicine. https://en.wikipedia.org/wiki/Medicine.
Health. https://en.wikipedia.org/wiki/Health.
Wehr, T. A. Photoperiodism in humans and other primates: evidence and implications. J. Biol. Rhythms 16, 348–364 (2001).
CAS PubMed Google Scholar
Roenneberg, T. & Aschoff, J. Annual rhythm of human reproduction: I. Biology, sociology, or both?. J. Biol. Rhythms 5, 195–216 (1990).
CAS PubMed Google Scholar
Roenneberg, T. & Merrow, M. The circadian clock and human health. Curr. Biol. 26, R432–R443 (2016).
CAS PubMed Google Scholar
Hotta, C. T. et al. Modulation of environmental responses of plants by circadian clocks. Plant Cell Environ. 30, 333–349 (2007).
CAS PubMed Google Scholar
Mattson, M. P. et al. Meal frequency and timing in health and disease. Proc. Natl. Acad. Sci. USA 111, 16647–16653 (2014).
CAS PubMed PubMed Central Google Scholar
Ruben, M. D. et al. A database of tissue-specific rhythmically expressed human genes has potential applications in circadian medicine. Sci. Transl. Med. 10, eaat8806 (2018).
PubMed PubMed Central Google Scholar
Chang, A.-M., Scheer, F. A. J. L. & Czeisler, C. A. The human circadian system adapts to prior photic history. J. Physiol. 589, 1095–1102 (2011).
CAS PubMed PubMed Central Google Scholar
Scheer, F. A. J. L., Wright, K. P., Kronauer, R. E. & Czeisler, C. A. Plasticity of the intrinsic period of the human circadian timing system. PLoS One 2, e721 (2007).
PubMed PubMed Central Google Scholar
Pittendrigh, C. S. & Daan, S. A functional analysis of circadian pacemakers in nocturnal rodents. I. The stability and lability of spontaneous frequency. J. Comp. Physiol. A. 106, 223–252 (1976).
Google Scholar
Brandstätter, R. Encoding time of day and time of year by the avian circadian system. J. Neuroendocrinol. 15, 398–404 (2003).
PubMed Google Scholar
Kramer, A. et al. Foundations of circadian medicine. PLoS Biol. 20, e3001567 (2022).
CAS PubMed PubMed Central Google Scholar
Roenneberg, T. Reflections of a conceptualist. NPJ Biol. Timing Sleep. 2, 2 (2025).
PubMed PubMed Central Google Scholar
Klerman, E. B. & St Hilaire, M. On mathematical modeling of circadian rhythms, performance, and alertness. J. Biol. Rhythms 22, 91–102 (2007).
PubMed Google Scholar
Roenneberg, T., Foster, R. & Klerman, E. The circadian system, sleep, and the health/disease balance: a conceptual review. J. Sleep. Res. 31, e13621 (2022).
PubMed PubMed Central Google Scholar
Muller, J. E. et al. Circadian variation in the frequency of onset of acute myocardial infarction. N. Engl. J. Med 313, 1315–1322 (1985).
CAS PubMed Google Scholar
Kaiser, I. H. & Halberg, F. Circadian periodic aspects of birth. Ann. N. Y Acad. Sci. 98, 1056–1068 (1962).
CAS PubMed Google Scholar
Wright, V. Some observations on diurnal variation of grip. Clin. Sci. 18, 17–23 (1959).
CAS PubMed Google Scholar
Saba, G. C., Materazzi, F. & Hoet, J. J. Diurnal rhythm of the adrenal cortical activity and of the cortisol metabolism in normal subjects. Folia Endocrinol. 16, 611–616 (1963).
CAS PubMed Google Scholar
Pickering, T. G. & Kario, K. Nocturnal non-dipping: what does it augur?. Curr. Opin. Nephrol. Hypertens. 10, 611–616 (2001).
CAS PubMed Google Scholar
Dashti, H. S. et al. Advancing chrononutrition for cardiometabolic health: a 2023 national heart, lung, and blood institute workshop report. JAHA 14, e039373 (2025).
PubMed PubMed Central Google Scholar
Ruben, M. D., Smith, D. F., FitzGerald, G. A. & Hogenesch, J. B. Dosing time matters. Science 365, 547–549 (2019).
CAS PubMed PubMed Central Google Scholar
Morris, C. J., Yang, J. N. & Scheer, F. A. J. L. The impact of the circadian timing system on cardiovascular and metabolic function. Prog. Brain Res. 199, 337–358 (2012).
CAS PubMed PubMed Central Google Scholar
Wittmann, M., Dinich, J., Merrow, M. & Roenneberg, T. Social jetlag: misalignment of biological and social time. Chronobiol. Int. 23, 497–509 (2006).
PubMed Google Scholar
Caliandro, R., Streng, A. A., Van Kerkhof, L. W. M., Van Der Horst, G. T. J. & Chaves, I. Social jetlag and related risks for human health: a timely review. Nutrients 13, 4543 (2021).
PubMed PubMed Central Google Scholar
Phillips, A. J. K. et al. Irregular sleep/wake patterns are associated with poorer academic performance and delayed circadian and sleep/wake timing. Sci. Rep. 7, 3216 (2017).
PubMed PubMed Central Google Scholar
Sletten, T. L. et al. The importance of sleep regularity: a consensus statement of the National Sleep Foundation sleep timing and variability panel. Sleep Health S2352721823001663 https://doi.org/10.1016/j.sleh.2023.07.016. (2023).
Roenneberg, T., Winnebeck, E. C. & Klerman, E. B. Daylight saving time and artificial time zones—a battle between biological and social times. Front. Physiol. 10, 944 (2019).
PubMed PubMed Central Google Scholar
James, F. O., Boivin, D. B., Charbonneau, S., Bélanger, V. & Cermakian, N. Expression of clock genes in human peripheral blood mononuclear cells throughout the sleep/wake and circadian cycles. Chronobiol. Int. 24, 1009–1034 (2007).
CAS PubMed Google Scholar
Doi, M. et al. Salt-sensitive hypertension in circadian clock-deficient Cry-null mice involves dysregulated adrenal Hsd3b6. Nat. Med. 16, 67–74 (2010).
CAS PubMed Google Scholar
Anea, C. B. et al. Increased superoxide and endothelial NO synthase uncoupling in blood vessels of Bmal1-knockout mice. Circ. Res. 111, 1157–1165 (2012).
CAS PubMed PubMed Central Google Scholar
Stow, L. R. et al. The circadian protein period 1 contributes to blood pressure control and coordinately regulates renal sodium transport genes. Hypertension 59, 1151–1156 (2012).
CAS PubMed PubMed Central Google Scholar
Richards, J., Diaz, A. N. & Gumz, M. L. Clock genes in hypertension: novel insights from rodent models. Blood Press Monit. 19, 249–254 (2014).
PubMed PubMed Central Google Scholar
Viswambharan, H. et al. Mutation of the circadian clock gene Per2 alters vascular endothelial function. Circulation 115, 2188–2195 (2007).
CAS PubMed Google Scholar
Marcheva, B. et al. Disruption of the clock components CLOCK and BMAL1 leads to hypoinsulinaemia and diabetes. Nature 466, 627–631 (2010).
CAS PubMed PubMed Central Google Scholar
Klerman, E. B. et al. Characterizing gun violence by time, day of the week, holidays, and month in 6 US Cities, 2015-2021. J. Biol. Rhythms 39, 100–108 (2024).
PubMed Google Scholar
Jones, S. S. et al. Forecasting daily patient volumes in the emergency department. Acad. Emerg. Med. 15, 159–170 (2008).
PubMed Google Scholar
Wang, T., Kaida, N. & Kaida, K. Effects of outdoor artificial light at night on human health and behavior: a literature review. Environ. Pollut. 323, 121321 (2023).
CAS PubMed Google Scholar
Martelli, M. et al. Artificial light at night disrupts fertility in Drosophila melanogaster. Comp. Biochem Physiol. C. Toxicol. Pharm. 299, 110349 (2026).
CAS Google Scholar
Yadav, V., Sharma, A., Tiwari, J. & Malik, S. Lost in the light: effects of exposure to artificial light at night on migratory birds. J. Exp. Zool. A Ecol. Integr. Physiol. https://doi.org/10.1002/jez.70043 (2025).
Article PubMed Google Scholar
Klerman, E. B., Kramer, A. & Zee, P. C. From bench to bedside and back again: translating circadian science to medicine. J. Biol. Rhythms 074873042211427 https://doi.org/10.1177/07487304221142743. (2022).
Cederroth, C. R. et al. Medicine in the fourth dimension. Cell Metab. 30, 238–250 (2019).
CAS PubMed PubMed Central Google Scholar
Allada, R. & Bass, J. Circadian mechanisms in medicine. N. Engl. J. Med. 384, 550–561 (2021).
CAS PubMed PubMed Central Google Scholar
Garaulet, M. et al. Timing of food intake predicts weight loss effectiveness. Int. J. Obes. 37, 604–611 (2013).
CAS Google Scholar
Allison, K. C. et al. Prolonged, controlled daytime versus delayed eating impacts weight and metabolism. Curr. Biol. 31, 650–657.e3 (2021).
CAS PubMed Google Scholar
Buttgereit, F. et al. Targeting pathophysiological rhythms: prednisone chronotherapy shows sustained efficacy in rheumatoid arthritis. Ann. Rheum. Dis. 69, 1275–1280 (2010).
CAS PubMed PubMed Central Google Scholar
Saito, Y., Yoshida, S., Nakaya, N., Hata, Y. & Goto, Y. Comparison between morning and evening doses of simvastatin in hyperlipidemic subjects. A double-blind comparative study. Arterioscler. Thromb. 11, 816–826 (1991).
CAS PubMed Google Scholar
Hrushesky, W. J. et al. High-dose intensity, circadian-timed doxorubicin and cisplatin adjuvant chemotherapy for bladder cancer. Cancer Treat. Rep. 71, 915–919 (1987).
CAS PubMed Google Scholar
Brunner-Ziegler, S. et al. Comparison between the impact of morning and evening doses of rivaroxaban on the circadian endogenous coagulation rhythm in healthy subjects. J. Thromb. Haemost. 14, 316–323 (2016).
CAS PubMed Google Scholar
Long, J. E. et al. Morning vaccination enhances antibody response over afternoon vaccination: a cluster-randomised trial. Vaccine 34, 2679–2685 (2016).
CAS PubMed PubMed Central Google Scholar
McCullar, K. S. et al. Timing of diuretic administration effects on urine volume in hospitalized patients. Front Physiol. 14, 1208324 (2023).
PubMed Google Scholar
Czeisler, C. A. et al. Modafinil for excessive sleepiness associated with shift-work sleep disorder. N. Engl. J. Med 353, 476–486 (2005).
CAS PubMed Google Scholar
Rajaratnam, S. M. et al. Melatonin agonist tasimelteon (VEC-162) for transient insomnia after sleep-time shift: two randomised controlled multicentre trials. Lancet 373, 482–491 (2009).
CAS PubMed Google Scholar
Lockley, S. W. et al. Tasimelteon for non-24-hour sleep–wake disorder in totally blind people (SET and RESET): two multicentre, randomised, double-masked, placebo-controlled phase 3 trials. Lancet 386, 1754–1764 (2015).
CAS PubMed Google Scholar
Morag, I. & Ohlsson, A. Cycled light in the intensive care unit for preterm and low birth weight infants. Cochr. Data. System. Rev. 2020, (2016).
Weiss, B. et al. Exposure to light and darkness and its influence on physiological measures of intensive care unit patients—a systematic literature review. Physiol. Meas. 37, R73–R87 (2016).
CAS PubMed Google Scholar
Aschoff, J. Exogenous and endogenous components in circadian rhythms. in Cold Spring Harbor Symposia on Quantitative Biology 11–28 (Cold Spring Harbor Press, 1960).
Brüning, F. et al. Sleep-wake cycles drive daily dynamics of synaptic phosphorylation. Science 366, eaav3617 (2019).
PubMed Google Scholar
Mrosovsky, N. Masking: history, definitions, and measurement. Chronobiol. Int. 16, 415–429 (1999).
CAS PubMed Google Scholar
Klerman, E. B. et al. Keeping an eye on circadian time in clinical research and medicine. Clin. Transl. Med. 12, (2022).
Ruben, M. D. et al. A large-scale study reveals 24-h operational rhythms in hospital treatment. Proc. Natl. Acad. Sci. USA 116, 20953–20958 (2019).
CAS PubMed PubMed Central Google Scholar
Schwartz, W. J., Reppert, S. M., Eagan, S. M. & Moore-Ede, M. C. In vivo metabolic activity of the suprachiasmatic nuclei: a comparative study. Brain Res. 274, 184–187 (1983).
CAS PubMed Google Scholar
Esposito, E. et al. Potential circadian effects on translational failure for neuroprotection. Nature 582, 395–398 (2020).
CAS PubMed PubMed Central Google Scholar
Joshi, D. et al. The association between clock gene polymorphisms and type 2 diabetes: a systematic review and meta-analysis. Diab. Metab. Syndr. 19, 103284 (2025).
CAS Google Scholar
Hiemstra, F. W. et al. Challenges and recommendations for integrating circadian medicine in critical care: a roadmap. Chest S0012-S3692(25)05931–8 https://doi.org/10.1016/j.chest.2025.12.010. (2025).
Bowman, C. et al. A method for characterizing daily physiology from widely used wearables. Cell Rep. Methods 1, 100058 (2021).
CAS PubMed PubMed Central Google Scholar
Phillips, N. E., Collet, T.-H. & Naef, F. Uncovering personalized glucose responses and circadian rhythms from multiple wearable biosensors with Bayesian dynamical modeling. Cell Rep. Methods 3, 100545 (2023).
CAS PubMed PubMed Central Google Scholar
Anafi, R. C., Francey, L. J., Hogenesch, J. B. & Kim, J. CYCLOPS reveals human transcriptional rhythms in health and disease. Proc. Natl. Acad. Sci. USA 114, 5312–5317 (2017).
CAS PubMed PubMed Central Google Scholar
Ueda, H. R. et al. Molecular-timetable methods for detection of body time and rhythm disorders from single-time-point genome-wide expression profiles. Proc. Natl. Acad. Sci. USA 101, 11227–11232 (2004).
CAS PubMed PubMed Central Google Scholar
Braun, R. et al. Universal method for robust detection of circadian state from gene expression. Proc. Natl. Acad. Sci. USA 115, E9247–E9256 (2018).
CAS PubMed PubMed Central Google Scholar
Wittenbrink, N. et al. High-accuracy determination of internal circadian time from a single blood sample. J. Clin. Investig. 128, 3826–3839 (2018).
PubMed PubMed Central Google Scholar
Kolodyazhniy, V. et al. Estimation of human circadian phase via a multi-channel ambulatory monitoring system and a multiple regression model. J. Biol. Rhythms 26, 55–67 (2011).
PubMed Google Scholar
Brown, L. S. et al. A classification approach to estimating human circadian phase under circadian alignment from actigraphy and photometry data. J. Pineal Res. 71, (2021).
Cain, S. W. et al. Sex differences in phase angle of entrainment and melatonin amplitude in humans. J. Biol. Rhythms 25, 288–296 (2010).
CAS PubMed PubMed Central Google Scholar
Duffy, J. F., Dijk, D. J., Klerman, E. B. & Czeisler, C. A. Later endogenous circadian temperature nadir relative to an earlier wake time in older people. Am. J. Physiol. 275, R1478–R1487 (1998).
CAS PubMed Google Scholar
Kantermann, T., Sung, H. & Burgess, H. J. Comparing the morningness-eveningness questionnaire and Munich chronotype questionnaire to the dim light melatonin onset. J. Biol. Rhythms 30, 449–453 (2015).
PubMed PubMed Central Google Scholar
Cox, R. C. et al. Distribution of dim light melatonin offset (DLMOff) and phase relationship to waketime in healthy adults and associations with chronotype. Sleep. Health 10, S76–S83 (2024).
PubMed Google Scholar
Gooley, J. J. et al. Exposure to room light before bedtime suppresses melatonin onset and shortens melatonin duration in humans. J. Clin. Endocrinol. Metab. 96, E463–E472 (2011).
CAS PubMed Google Scholar
Wright, K. P., Gronfier, C., Duffy, J. F. & Czeisler, C. A. Intrinsic period and light intensity determine the phase relationship between melatonin and sleep in humans. J. Biol. Rhythms 20, 168–177 (2005).
PubMed PubMed Central Google Scholar
Wright, K. P. et al. Entrainment of the human circadian clock to the natural light-dark cycle. Curr. Biol. 23, 1554–1558 (2013).
CAS PubMed PubMed Central Google Scholar
Rahman, S. A., Kayumov, L., Tchmoutina, E. A. & Shapiro, C. M. Clinical efficacy of dim light melatonin onset testing in diagnosing delayed sleep phase syndrome. Sleep. Med. 10, 549–555 (2009).
PubMed Google Scholar
Flynn-Evans, E. E. et al. Circadian phase and phase angle disorders in primary insomnia. Sleep 40, (2017).
Brown, L. S., Klerman, E. B. & Doyle, F. J. An anticipatory scheme for the model predictive control of circadian phase for expected environmental light changes. IEEE Control Syst. Lett. 1–1 https://doi.org/10.1109/LCSYS.2021.3129475. (2021).
Abel, J. H., Chakrabarty, A., Klerman, E. B. & Doyle, F. J. 3rd Pharmaceutical-based entrainment of circadian phase via nonlinear model predictive control. Automatica 100, 336–348 (2019).
PubMed Google Scholar
Amendolara, B., Miller, L., Samudrala, S. & McCarthy, M. J. Morning-evening differences in the post-illumination pupillary response are associated with seasonal mood changes. Chronobiol. Int. 42, 520–527 (2025).
PubMed Google Scholar
Vlasac, I. M. et al. A novel home-based study of circadian rhythms: design, rationale, and methods for the circadia study. Sleep zsad197 https://doi.org/10.1093/sleep/zsad197. (2023).
Klerman, E. B., Wang, W., Phillips, A. J. K. & Bianchi, M. T. Statistics for sleep and biological rhythms research. J. Biol. Rhythms 32, 18–25 (2017).
PubMed Google Scholar
Leise, T. L. Analysis of nonstationary time series for biological rhythms research. J. Biol. Rhythms 32, 187–194 (2017).
CAS PubMed Google Scholar
International Agency for Research on Cancer. Painting, Firefighting, and Shiftwork. (2009).
American Academy of Sleep Medicine. International Classification of Sleep Disorders. (American Academy of Sleep Medicine (AASM, 2014).
American Heart Association. Life’s Essential Eight. https://www.heart.org/en/healthy-living/healthy-lifestyle/lifes-essential-8 (2025).
Brainard, G. C., Barger, L. K., Soler, R. R. & Hanifin, J. P. The development of lighting countermeasures for sleep disruption and circadian misalignment during spaceflight. Curr. Opin. Pulm. Med. 22, 535–544 (2016).
PubMed Google Scholar
Tubbs, A. S., Fernandez, F.-X., Grandner, M. A., Perlis, M. L. & Klerman, E. B. The mind after midnight: nocturnal wakefulness, behavioral dysregulation, and psychopathology. Front. Netw. Physiol. 1, 830338 (2022).
PubMed PubMed Central Google Scholar
Malecki, K. M. C. et al. Integrating environment and aging research: opportunities for synergy and acceleration. Front Aging Neurosci. 14, 824921 (2022).
CAS PubMed PubMed Central Google Scholar
Danziger, S., Levav, J. & Avnaim-Pesso, L. Extraneous factors in judicial decisions. Proc. Natl. Acad. Sci. USA 108, 6889–6892 (2011).
CAS PubMed PubMed Central Google Scholar
McAlpine, C. S. et al. Sleep modulates haematopoiesis and protects against atherosclerosis. Nature 566, 383–387 (2019).
CAS PubMed PubMed Central Google Scholar
Download references
Discussions with Dr. Patricia Carter, Dr. Melissa Knauert, Dr. Till Roenneberg, Dr. Norah Simpson. Manuscript review by Dr. Jennifer Grom and Dr. Daniel Blum.
Department of Neurology, MassGeneralBrigham, Boston, MA, USA
Elizabeth B. Klerman
Division of Sleep and Circadian Disorders, Brigham and Women’s Hospital, Boston, MA, USA
Elizabeth B. Klerman
Division of Sleep Medicine, Harvard Medical School, Boston, MA, USA
Elizabeth B. Klerman
PubMed Google Scholar
E.B.K.: ideas, literature review, writing, editing.
Correspondence to Elizabeth B. Klerman.
E.B.K.: (i) travel: Santa Fe Institute, Sleep Research Society (SRS), Society for Research on Biological Rhythms (SRBR), Lorentz Center (the Netherlands); (ii) honoraria: SRS; (iii) consulting: Buck Institute on Aging, Circadian Therapeutics, Guidepoint; (iv) scientific advisory board (unpaid): Chronsulting: (v) other: Partner is founder and CEO of Chronsulting.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
Reprints and permissions
Klerman, E.B. Translational applications of circadian research: connecting chronobiology to medicine. npj Biol Timing Sleep 3, 18 (2026). https://doi.org/10.1038/s44323-026-00084-2
Download citation
Received:
Accepted:
Published:
Version of record:
DOI: https://doi.org/10.1038/s44323-026-00084-2
Anyone you share the following link with will be able to read this content:
Sorry, a shareable link is not currently available for this article.
Provided by the Springer Nature SharedIt content-sharing initiative
Collection
Advertisement
npj Biological Timing and Sleep (npj Biol Timing Sleep)
ISSN 2948-281X (online)
© 2026 Springer Nature Limited
Sign up for the Nature Briefing newsletter — what matters in science, free to your inbox daily.